Filling out the Florida 3170 form can be a complex task, and many applicants make common mistakes that can delay their application process. One frequent error is failing to submit the application at least 60 days before the current license expires. This requirement is crucial to avoid incurring late fees. Applicants should always mark their calendars to ensure timely submission.
Another mistake involves neglecting to include all required documents and fees. The application will be withdrawn from review if any essential materials are missing. It is advisable to double-check the checklist provided with the form to ensure completeness before submission.
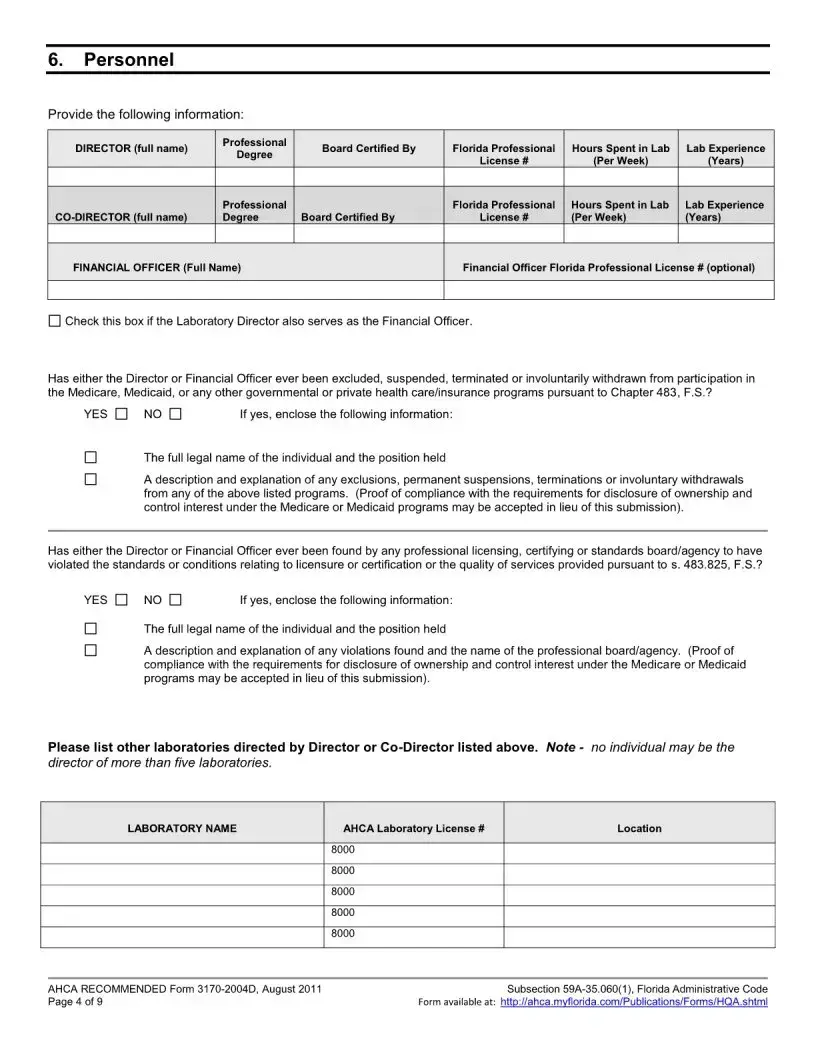
Some applicants overlook the importance of the Level 2 background screening for both the Lab Director and Financial Officer. This screening must be conducted every five years, and the results should be sent directly to the Agency for Health Care Administration. Failing to provide this documentation can lead to unnecessary delays.
Using incorrect payment methods is another common issue. The form specifies that starter checks and temporary checks are not accepted. Therefore, applicants must ensure they use a check or money order that complies with this requirement to avoid processing issues.
Many people also make the mistake of not providing sufficient evidence of the director's qualifications. Documentation must clearly demonstrate the necessary laboratory experience and training. If this is not adequately shown, the application may be rejected.
Some applicants mistakenly submit fingerprint cards without ensuring they are filled out completely. Law enforcement personnel or individuals trained in processing fingerprints must take the fingerprints. Incomplete cards can lead to further complications in the application process.
Another common error is failing to use the correct LiveScan vendor. If an applicant chooses a LiveScan source other than the Agency’s contracted vendor, they must provide specific identification details to ensure proper processing. Not doing so can result in additional screening requirements and fees.
In addition, many applicants do not include the Affidavit of Compliance with Background Screening Requirements. This affidavit is essential and must accompany the application to confirm that the required screenings have been conducted.
Some individuals fail to follow the instructions regarding the organization of their documents. The Agency requests that checks, money orders, and fingerprint cards be placed on top of the application and paperclipped together. Stapling or binding the documents can complicate the scanning process.
Finally, some applicants do not take the time to read the definitions provided in the application. Understanding terms such as "Licensee" and "Provider" is vital for accurately completing the form. Misinterpretation of these terms can lead to mistakes that affect the application’s success.