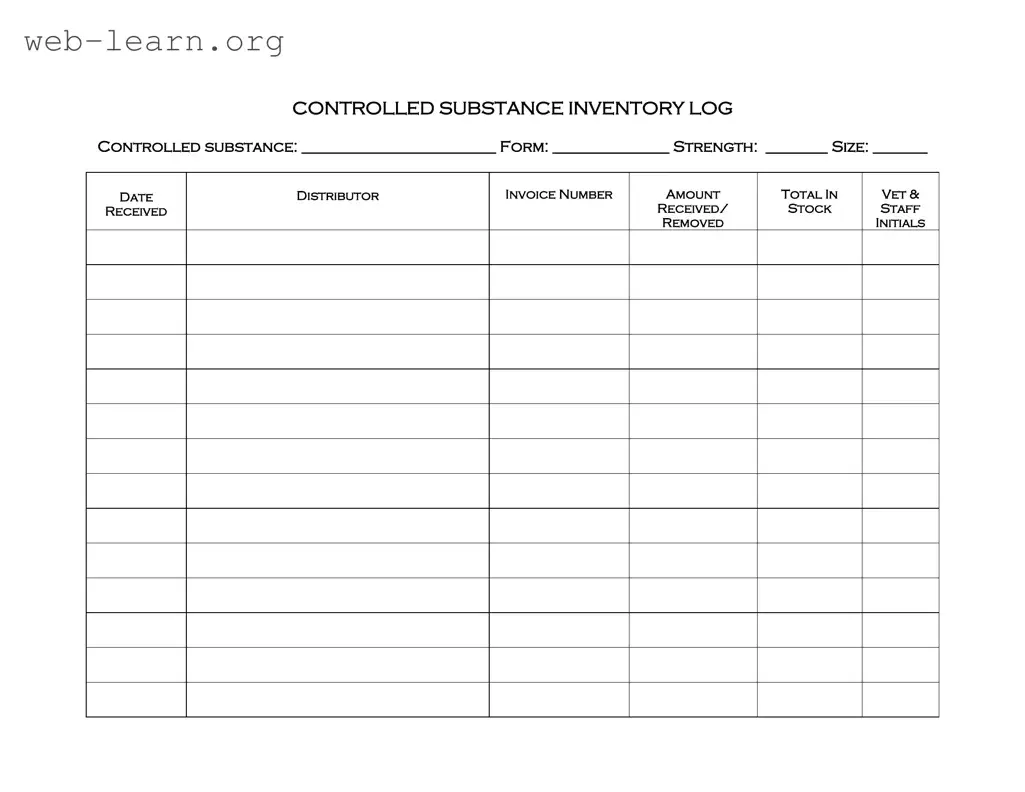
The Controlled Drug Log form serves as a critical tool for managing and tracking the use of controlled substances within various settings, particularly in veterinary practices. This comprehensive log includes essential fields that ensure accurate documentation of each controlled substance, such as the name of the drug, its specific form, strength, and size. Additionally, the form requires the date of receipt, distributor information, and invoice number, which help maintain a clear chain of custody. Users must also record the amount received and the total stock available, allowing for effective inventory management. Each entry is completed with the initials of the staff member responsible for the transaction, promoting accountability and transparency in handling these sensitive materials. By utilizing the Controlled Drug Log form, organizations can adhere to regulatory requirements while ensuring the safe and responsible use of controlled substances.